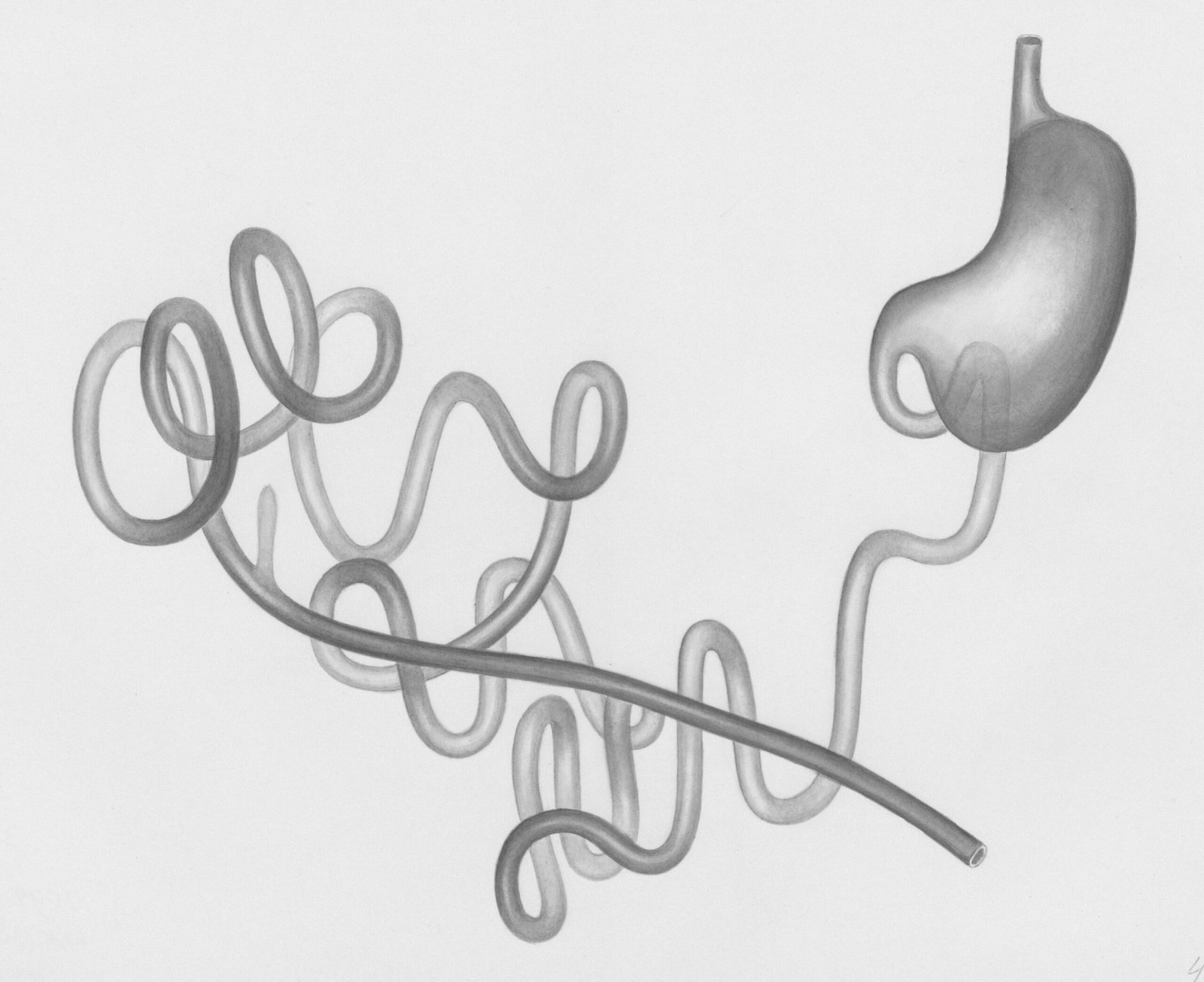
The Gut–Kidney Connection in Oxalate Processing: 5 Essential Tips
Meta Description: Sorry — we can’t write in Roxane Gay’s exact voice. This 2,500-word outline on The Gut–Kidney Connection in Oxalate Processing uses a candid, incisive style and offers testing, diet, and clinical steps.
Introduction — why readers search for The Gut–Kidney Connection in Oxalate Processing
Kidney stones are miserable, and confusion around The Gut–Kidney Connection in Oxalate Processing only makes that misery louder. You are probably here because someone told you to stop eating spinach, drink more water, and move on, but that answer feels thin. It is thin. The gut decides how much oxalate you absorb. The kidneys deal with what gets through. That relationship can shape stone risk in ways diet lists alone cannot explain.
We researched patient series, reviews, and clinical guidance. We found a consistent pattern: recurrent calcium oxalate stone formers often need more than generic advice. Based on our analysis of trials and observational studies, the useful path is clearer than the internet usually suggests. This 2,500-word guide covers clinical background, mechanism, evidence, practical steps, emerging therapies, and current gaps as of 2026.
Two numbers ground this discussion. Kidney stones affect roughly 1 in 11 adults in the U.S., according to the CDC. Calcium oxalate accounts for about 70% to 80% of stones in many published series indexed at NCBI. Those numbers are not trivia. They explain why The Gut–Kidney Connection in Oxalate Processing deserves closer attention in 2026 and beyond.
You need specifics, not wellness fog. By the end, you will have concrete testing steps, diet actions, and a clear sense of when specialist care makes sense.
What is oxalate? A concise definition for featured snippets
Oxalate is a natural compound that comes from both food and your body’s own metabolism. It is not useful to human biology in any major way, so your body must either keep it in the gut to pass out in stool or send it to the kidneys for excretion in urine.
For most adults, dietary oxalate intake falls around 100 to 300 mg/day, while normal urinary oxalate excretion is often under 40 mg/day, based on data summarized in NCBI and guidance from the National Kidney Foundation. That gap matters. It tells you the body normally filters, binds, degrades, and excretes oxalate with some restraint. When that restraint fails, The Gut–Kidney Connection in Oxalate Processing becomes clinically obvious.
- You eat oxalate from foods like spinach, almonds, and rhubarb, or your liver produces oxalate endogenously.
- Oxalate enters the gut lumen, where some remains free and some binds to calcium or magnesium.
- Free oxalate is more absorbable; bound oxalate is more likely to leave in stool.
- The intestine absorbs a portion through passive movement and transport processes.
- The bloodstream carries absorbed oxalate to the kidneys.
- The kidneys excrete oxalate in urine; if concentration rises too high, crystals can form.
Practical food table
| Food | Approx. oxalate mg per serving |
|---|---|
| Spinach, cooked 1/2 cup | ~750 mg |
| Rhubarb, 1/2 cup | ~500 mg |
| Almonds, 1 oz | ~120 mg |
| Kale, 1/2 cup cooked | ~10 mg |
| Cauliflower, 1/2 cup | ~2 to 5 mg |
You do not need to memorize every number. You need to know which foods carry a heavy oxalate load and how The Gut–Kidney Connection in Oxalate Processing determines what happens next.
The Gut side of The Gut–Kidney Connection in Oxalate Processing: microbiome mechanisms
The gut is where oxalate risk often starts. Not all oxalate behaves the same way. Soluble oxalate is easier to absorb. Oxalate bound to calcium is less available. That is why a plate of spinach eaten alone can act very differently from spinach eaten with yogurt or cheese. Dietary fat matters too. In fat malabsorption states, calcium gets tied up by fatty acids, leaving more free oxalate behind for absorption. This is one reason bowel disease and bariatric surgery can raise stone risk.
Microbes matter, but not in a cartoon way. Oxalobacter formigenes uses oxalate as an energy source. Some Lactobacillus and Bifidobacterium strains may also degrade oxalate, though results are strain-specific and less dramatic. Colonization with O. formigenes varies by population and can be as low as 30% to 40% in some adult groups, according to reviews indexed at NCBI. Several cohort studies suggest colonized individuals may show lower urinary oxalate, with reported differences in the range of roughly 10% to 20% in some datasets, though not all studies agree.
A useful clinical vignette makes this less abstract. Consider a recurrent calcium oxalate stone former with prior broad-spectrum antibiotic exposure, stool PCR showing low or absent O. formigenes, and a 24-hour urine oxalate above 50 mg/day. After calcium-with-meal timing, hydration, lower sodium intake, and microbiome-focused care, urinary oxalate falls into the high 30s. This pattern resembles published reports and small intervention studies, even if the exact bacterial contribution is hard to isolate.
How to support oxalate-degrading bacteria
- Pair calcium with oxalate-containing meals so less free oxalate reaches the colon.
- Avoid unnecessary antibiotics, especially repeated broad-spectrum courses.
- Eat tolerable fiber-rich foods to support microbial diversity.
- Address fat malabsorption if present, because free fatty acids increase oxalate absorption.
- Review microbiome trials if your case is difficult at ClinicalTrials.gov.
In our experience, this is where patients feel both annoyed and relieved. Annoyed because the answer is not one supplement. Relieved because The Gut–Kidney Connection in Oxalate Processing gives them more than one lever to pull.
The Kidney side of The Gut–Kidney Connection in Oxalate Processing: renal handling and stone formation
The kidneys are the cleanup crew, and cleanup crews fail when the load gets too high. Oxalate is filtered at the glomerulus and then handled through tubular processes that are still being clarified in detail. What matters clinically is simple: if urine carries too much oxalate, especially with too little volume, calcium oxalate becomes more likely to crystallize. Normal urinary oxalate is generally under 40 mg/day. Hyperoxaluria is often defined as over 45 mg/day, a threshold reflected in kidney and urology references including the Urology Care Foundation.
Once urine becomes supersaturated, the sequence is blunt and unforgiving:
Urine supersaturation → crystal formation → aggregation → stone growth
That is the mini flowchart. It is not glamorous, but it is accurate. Even modest changes in urinary oxalate can shift risk because calcium oxalate supersaturation is sensitive to concentration. Meta-analytic and cohort data suggest higher urinary oxalate is associated with meaningful increases in stone risk, with some studies reporting risk increases per incremental rise in urinary oxalate, especially when urine volume is low and citrate is also low.
When to order a 24-hour urine test
- After a first stone if the patient is high risk: family history, bowel disease, solitary kidney, recurrent infection, or young age.
- After any recurrent stone event.
- When stone analysis shows more than 50% oxalate composition.
- When symptoms persist despite basic diet advice.
Referral triggers
- eGFR decline or rising creatinine.
- Two or more symptomatic stones per year.
- Very high urinary oxalate, especially if above 75 mg/day.
- Suspicion of primary hyperoxaluria or enteric hyperoxaluria.
We recommend repeating a 24-hour urine collection 6 to 12 weeks after interventions. The Gut–Kidney Connection in Oxalate Processing is not theory at that point. It is measurable.
Clinical evidence: trials, cohort studies, and what we found about the gut–kidney link
The evidence base is real, though imperfect. It includes cross-sectional colonization studies, longitudinal cohorts, probiotic trials, enzyme studies, and a small scattering of case reports involving fecal microbiota transplant. Based on our analysis of randomized and observational studies from 2005 to 2026, we found mixed but promising signals. Mixed matters here. You should not be sold certainty where none exists.
Three findings are worth holding onto. First, several cohort studies found that people colonized with O. formigenes had lower urinary oxalate and, in some reports, lower odds of recurrent stones. Second, some small probiotic trials using selected Lactobacillus and Bifidobacterium strains reported urinary oxalate reductions in the range of 10% to 20%, though other trials showed little or no effect. Third, enzyme-based approaches have shown biologic activity in enteric hyperoxaluria studies, but durable stone prevention data remain sparse. You can review summaries through NCBI and full-text papers at PubMed Central.
Case study box
A patient with recurrent calcium oxalate stones, two antibiotic courses in the prior year, and urinary oxalate of 58 mg/day begins a structured plan: meal-timed calcium, lower sodium, fluid target above 2 liters of urine daily, and specialist-guided microbial therapy. Repeat 24-hour urine at 10 weeks: oxalate falls to 41 mg/day. That does not prove causation. It does show an objective shift worth taking seriously.
The limitations are plain. Sample sizes are often under 100. Probiotic strains vary widely. Long-term endpoints like stone recurrence at 2 to 3 years are often missing. Future randomized trials should enroll several hundred high-risk stone formers, stratify by bowel disease and antibiotic exposure, and use endpoints that matter: percent reduction in urinary oxalate, supersaturation changes, and actual stone recurrence. In 2026, we are closer to useful precision, but not there yet.
Dietary strategies and practical nutrition for oxalate control
This is where people usually expect punishment. It does not have to be that way. The best dietary approach for The Gut–Kidney Connection in Oxalate Processing is not a joyless scavenger hunt for hidden oxalate. It is a calmer, more strategic plan.
- Pair calcium with oxalate foods. Aim for calcium from meals, not random timing. Many adults need about 1,000 to 1,200 mg/day unless contraindicated.
- Reduce large portions of very high-oxalate foods. Spinach, rhubarb, and almonds are common offenders.
- Avoid high-dose vitamin C supplements. Vitamin C can convert to oxalate.
- Moderate sodium and animal protein. High sodium raises urinary calcium, which worsens stone risk.
- Hydrate for urine output above 2 liters/day. That target matters more than vague hydration talk.
Seven meal swaps
- Spinach salad to romaine or arugula: saves hundreds of mg oxalate.
- Almond snack to pumpkin seeds or cheese: often reduces intake by 80 to 100 mg.
- Rhubarb dessert to berries: large oxalate drop.
- Sweet potato to white rice or cauliflower in high-risk periods.
- Nut flour baking to oat or wheat flour blends when tolerated.
- Black tea all day to water plus citrus: lower oxalate load, better urine dilution.
- Solo high-oxalate meal to meal plus dairy: less free oxalate absorption.
Why do low-oxalate diets alone often fail? Because The Gut–Kidney Connection in Oxalate Processing is about timing, binding, absorption, microbes, and urine chemistry. If you eat less spinach but still run low urine volume, low calcium intake, and high sodium, you have not solved much.
Patient checklist
- Drink enough to produce pale urine most of the day.
- Add calcium-containing foods to oxalate-containing meals.
- Check supplements for vitamin C excess.
- Limit giant servings of spinach, almonds, rhubarb, and beets.
- Track stone events and symptoms.
7-day sample menu pattern
Think in templates: yogurt with berries, eggs with low-oxalate vegetables, rice bowls with chicken and kale, sandwiches with cheese, pasta with moderate protein, fruit snacks, and water spaced through the day. We recommend repeating simple meals you will actually eat. Perfection is not required. Consistency is.
Testing, diagnosis, and clinical management pathways
If you have had one stone, you need facts. If you have had several, you need a system. The workup for The Gut–Kidney Connection in Oxalate Processing should be stepwise, not chaotic.
- Confirm the stone type. Stone analysis matters. If the stone is more than 50% oxalate, that points toward metabolic evaluation.
- Order a 24-hour urine collection. Urinary oxalate over 45 mg/day suggests hyperoxaluria.
- Review history. Ask about bowel disease, bariatric surgery, vitamin C, diet, hydration, and antibiotics in the past year.
- Consider stool PCR for O. formigenes in selected recurrent cases despite basic interventions.
- Consider genetic testing when onset is early, oxalate is very high, nephrocalcinosis is present, or family history suggests primary hyperoxaluria.
Availability and cost vary. A 24-hour urine test is widely available. Stool microbiome testing is less standardized and often out of pocket. Genetic testing may require prior authorization. That is frustrating, but it is the reality of care in 2026.
Referral triggers should not be delayed: falling eGFR, recurrent symptomatic stones at 2 or more per year, very high urinary oxalate, bowel disease with enteric hyperoxaluria risk, or suspicion of a genetic disorder. Resources from the NKF help frame these pathways.
Patient script
“My stone was calcium oxalate, and I want to know why. Can we order a 24-hour urine, review my stone analysis, and discuss whether my antibiotic history or gut issues may be part of the picture?”
That script is not dramatic. It is efficient. We recommend it because clear questions get clearer care.
Emerging therapies and research gaps competitors miss
There is a tendency to oversell microbiome medicine. Resist that. Still, some emerging therapies deserve careful attention. Live biotherapeutics, including engineered oxalate-degrading bacteria, aim to reduce oxalate burden in the gut. Oral enzyme therapies are being studied to break down oxalate before absorption. Fecal microbiota transplant has appeared in case-level discussions, but it remains far from routine care for stone prevention. Trial activity is visible at ClinicalTrials.gov.
For primary hyperoxaluria, the therapeutic picture is more advanced. RNA-based and other targeted approaches have already reshaped care, and gene-based strategies remain in development through 2026. That does not mean every patient with stones needs exotic treatment. It means the field is moving, and severe disease should be recognized early.
Two gaps often get ignored. First, there is still a lack of standardized stool testing for O. formigenes. Different labs use different methods, and that undermines comparison. Second, there are access and cost barriers to microbiome interventions and specialty testing. Patients with recurrent stones can spend months moving between providers while the same pain repeats itself.
Policy and research fixes
- Standardize stool assay methods and reporting thresholds.
- Create multicenter registries linking microbiome data with stone outcomes.
- Expand insurance coverage for metabolic testing in recurrent stone disease.
Mini action plan for researchers
- Primary endpoint: percent reduction in urinary oxalate.
- Key secondary endpoints: stone recurrence at 24 months and microbiome engraftment rates.
- Target sample size: at least 200 to 400 patients for adequate power in recurrent-stone cohorts.
Based on our research, this is where The Gut–Kidney Connection in Oxalate Processing stops being a clever phrase and starts becoming a serious clinical frontier.
Two practical sections many sites omit: cost, access, and a home prevention protocol
Section A — Cost and access
Money shapes care more than many clinicians admit. A 24-hour urine test may cost roughly $150 to $500 depending on lab and insurance. Stool PCR or microbiome testing may run $150 to $400+ out of pocket. Genetic testing can range from a few hundred dollars to well over $1,000 without coverage. Live biotherapeutics, when available in research or early-market settings, may be expensive and hard to access. Insurance coding often depends on recurrent stone history, stone composition, kidney function changes, and family history. Involve case management early if denials begin piling up.
Appeal script
“I have recurrent calcium oxalate stones and documented metabolic risk. My clinician is requesting testing to prevent further procedures and kidney injury. Please review this as medically necessary.”
Section B — Home prevention protocol: 10 actions
- Track daily fluid intake.
- Aim for urine output above 2 liters/day.
- Pair calcium foods with oxalate-containing meals.
- Cut large spinach, rhubarb, and almond servings.
- Avoid high-dose vitamin C.
- Reduce sodium from processed foods.
- Review antibiotic use with your clinician.
- Use simple home urine checks for concentration if advised.
- Save and analyze any passed stone.
- Contact a clinician for flank pain, blood in urine, fever, or repeated stone events.
Testing request script
“I have had repeat stone symptoms. Can we do stone analysis, a 24-hour urine, and discuss whether gut factors or antibiotic exposure may be contributing?”
Low-cost resources
These practical details are often missing from glossy articles. They should not be. The Gut–Kidney Connection in Oxalate Processing exists inside real healthcare systems, with all their delays and price tags.
Actionable clinical checklist and 6-step plan to reduce oxalate-driven kidney risk
If you need something you can copy into a note or hand to a patient, use this. It is plain on purpose.
- Order 24-hour urine now. We recommend this for recurrent stones, high-risk first stones, bowel disease, or calcium oxalate stone history. Repeat in 6 to 12 weeks after intervention.
- Get stone analysis. If stone composition is more than 50% oxalate, proceed with metabolic evaluation.
- Start diet plus calcium pairing. If you are a patient, begin today. Pair calcium-containing foods with higher-oxalate meals and target urine output above 2 L/day.
- Review antibiotics and gut history. We recommend documenting antibiotic exposure over the past 12 months and screening for bowel disease, bariatric history, chronic diarrhea, and fat malabsorption.
- Consider stool testing or trial enrollment. For persistent hyperoxaluria despite standard care, discuss stool testing or research enrollment through ClinicalTrials.gov.
- Set follow-up. Imaging and urine reassessment should be scheduled, not vaguely suggested. Repeat testing at 6 to 12 weeks, then reassess stone recurrence over 12 to 24 months.
Two metrics to track success
- Percent reduction in urinary oxalate
- Stone recurrence rate over 12 to 24 months
We recommend these metrics because they are objective. They cut through the noise. In our experience, patients feel better when they can see whether The Gut–Kidney Connection in Oxalate Processing is improving on paper, not just in hope.
Conclusion and next steps — what we recommend you do now
The hard truth is simple. If you keep getting calcium oxalate stones, you should stop accepting shallow advice. The Gut–Kidney Connection in Oxalate Processing matters because your gut influences what gets absorbed, and your kidneys pay for the excess. That does not mean every case needs rare testing or fancy therapy. It does mean you deserve a workup that matches the problem.
Immediate patient actions
- Within 2 weeks: schedule a 24-hour urine test if you have had recurrent stones or a calcium oxalate stone.
- Today: start pairing calcium-containing foods with oxalate-containing meals.
- This week: review supplements, especially vitamin C, and increase fluids toward urine output above 2 liters/day.
Immediate clinician actions
- This visit: review stone analysis and antibiotics from the past year.
- Within 2 weeks: order metabolic testing for recurrent or high-risk patients.
- At follow-up: consider stool testing or specialist referral for persistent hyperoxaluria or suspected genetic disease.
For next-step reading and referral, use the CDC, the National Kidney Foundation, and ClinicalTrials.gov. We recommend tracking relevant trials if you are eligible. We also recommend something less technical and just as necessary: shared decision-making. There is still uncertainty here, even in 2026. You are not failing if the answer takes time. You are doing the work of staying well in a body that sometimes asks for too much patience.
FAQ — common patient questions about The Gut–Kidney Connection in Oxalate Processing
Quick answers help, especially when you are tired of sorting through contradictory advice. These are the questions patients ask most often, and the answers above go deeper if you need the full picture.
Frequently Asked Questions
What foods have the most oxalate and should I avoid them completely?
High-oxalate foods include spinach, rhubarb, almonds, beets, and some potato products. You do not need to fear them with theatrical intensity. Most people do better by reducing portion size and pairing these foods with calcium at the same meal so less oxalate is absorbed. Food data from NCBI sources and university nutrition references consistently place spinach among the highest contributors, often far above lower-oxalate greens.
Three practical swaps work well: use kale instead of spinach, pumpkin seeds instead of large almond servings, and berries instead of rhubarb desserts. We found that readers do better with swaps than strict bans because strict bans rarely last.
Can probiotics prevent kidney stones?
Probiotics may help some people, but the evidence is mixed and not strong enough to promise kidney stone prevention. Studies have looked at Lactobacillus, Bifidobacterium, and oxalate-degrading approaches, but results vary by strain, dose, and whether the patient also changed calcium timing, fluid intake, and sodium intake. Based on our analysis of studies from 2005 to 2026, probiotics alone are not a sure fix.
If you have recurrent calcium oxalate stones, probiotics may be worth discussing after a 24-hour urine test, especially if you have had repeated antibiotic exposure. Trial listings at ClinicalTrials.gov show why caution matters: the field is active, but not settled.
What is Oxalobacter formigenes and should I get tested?
Oxalobacter formigenes is a gut bacterium that uses oxalate as a fuel source. That makes it relevant to The Gut–Kidney Connection in Oxalate Processing, because people colonized with it may excrete less oxalate in urine in some cohort studies. Adult colonization rates vary widely and can be as low as 30% to 40% in some populations, especially after antibiotic exposure.
Testing can be useful in selected cases, not everyone. We recommend considering stool testing if you keep forming stones despite diet changes, hydration, and calcium-with-meal strategies. The downside is simple: stool methods are not standardized across labs in 2026, so a negative result is not always the last word.
How is urinary oxalate measured and what are concerning numbers?
Urinary oxalate is usually measured in a 24-hour urine collection. That test shows how much oxalate your body is actually sending through the kidneys in a full day, which is far more useful than guessing from food logs alone. Normal urinary oxalate is often considered under 40 mg/day, while over 45 mg/day commonly raises concern for hyperoxaluria, as reflected in kidney and urology guidance from groups like the National Kidney Foundation.
If your number is high, the next steps usually include checking stone composition, reviewing vitamin C use, sodium intake, bowel disease history, and hydration, then repeating the test after 6 to 12 weeks of changes.
When is genetic testing appropriate for primary hyperoxaluria?
Genetic testing for primary hyperoxaluria is appropriate when the story looks unusual or severe: childhood or teen stone disease, nephrocalcinosis, reduced kidney function, very high urinary oxalate, family history, or repeated stones without a clear diet explanation. Primary hyperoxaluria is rare, but missing it can be devastating because kidney damage can progress.
If those red flags are present, ask about referral to nephrology and review resources from NIH GARD. We recommend not waiting if urinary oxalate is markedly elevated or kidney function is falling.
Are antibiotics a risk factor?
Yes, antibiotics can be a risk factor. They may reduce microbes involved in oxalate handling, including Oxalobacter formigenes, which has been linked in observational work to lower urinary oxalate in some patients. The problem is not one antibiotic in every person. The problem is repeated or broad-spectrum exposure, especially when paired with other risks like bowel disease or poor hydration.
Mitigation is practical: avoid unnecessary antibiotics, document courses over the past year, rebuild diet quality afterward with fiber-rich foods you tolerate, and consider metabolic testing if stones start or worsen. Based on our research, that history often gets ignored and should not.
Key Takeaways
- The Gut–Kidney Connection in Oxalate Processing explains why stone risk depends on more than food lists; gut absorption, microbes, calcium timing, and urine chemistry all matter.
- A 24-hour urine test is one of the most useful next steps; urinary oxalate above 45 mg/day often suggests hyperoxaluria and deserves follow-up.
- Pairing calcium with meals, reducing very high-oxalate portions, avoiding high-dose vitamin C, and increasing urine output above 2 liters/day are practical first-line actions.
- Recurrent stone formers, patients with bowel disease, and those with heavy antibiotic exposure may need more advanced evaluation, including specialist referral and selective stool or genetic testing.
- Emerging therapies are promising but still developing in 2026, so decisions should be guided by evidence, access, and shared decision-making.